Protocol
The GeoSmoking Study investigates the causal effects of exposure to tobacco retail environments on smoking behavior and neural reactivity among daily cigarette smokers.
Study Setting and Design
🏛️ Study Overview
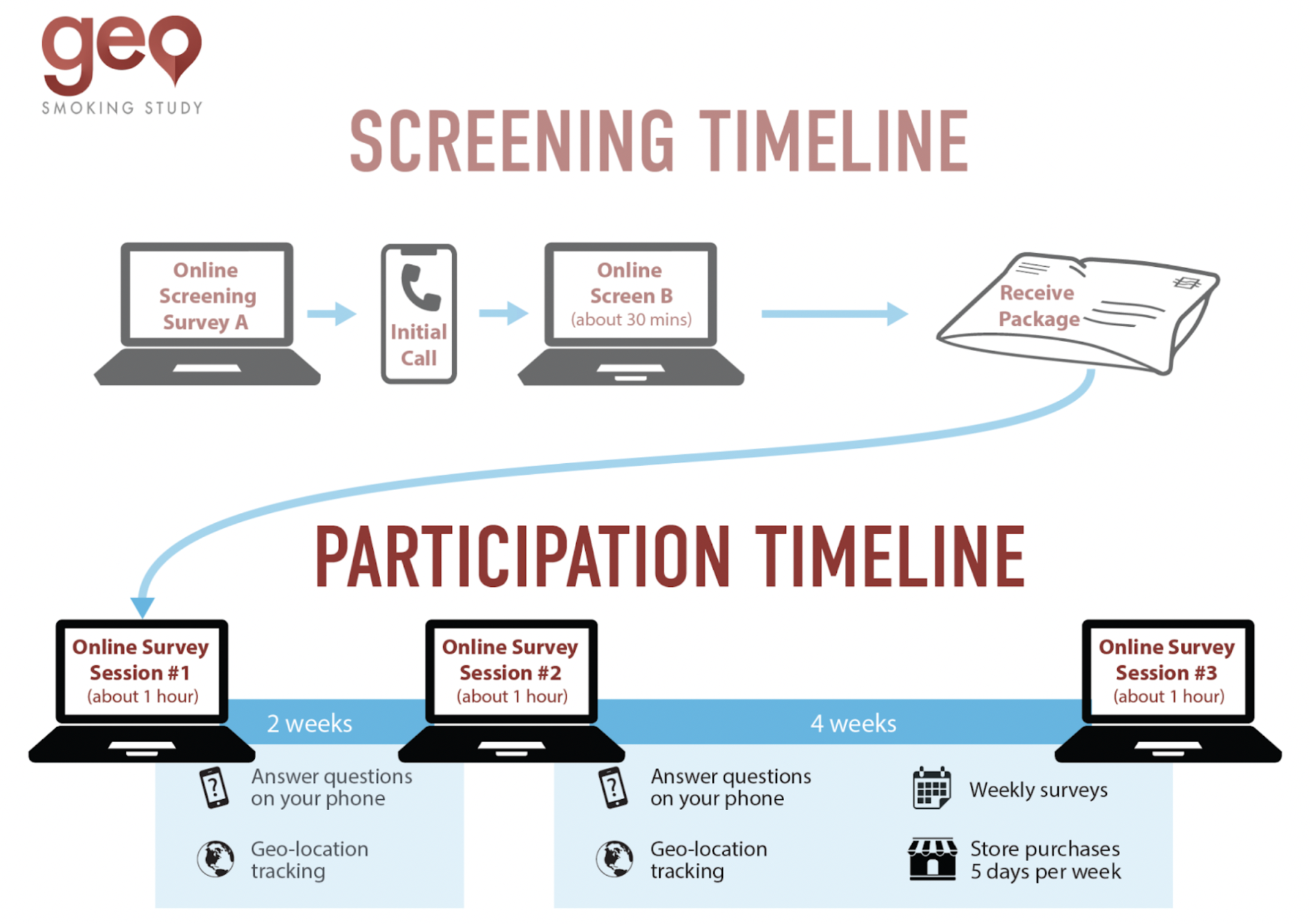
The GeoSmoking Study was conducted at the University of Pennsylvania. Adults who smoked cigarettes daily were recruited across PA, DE, and NJ to complete a multi-part remote study.
The study consisted of three main components:
Baseline Period
2 weeks - EMA reporting, geolocation tracking, tobacco retail exposure assessment
Intervention Period
4 weeks - Randomized store visits, continued EMA and geolocation tracking
fMRI Session
Optional - On-campus neural reactivity assessment
During the two-week baseline period, participants reported craving and smoking via EMA (Ecological Momentary Assessment) using the LifeData RealLife Exp app multiple times per day while their geolocation was tracked to calculate tobacco retail exposure. The following four-week intervention period was designed to assess causal effects of exposure to tobacco retail.
Participants were randomly assigned to enter a tobacco retail store or a non-tobacco retail store once per day, or to follow their normal routines, while EMA and geolocation continued to be collected. Following the intervention period, eligible participants could opt into a fMRI session conducted at the University of Pennsylvania, designed to investigate neural reactivity to tobacco marketing cues.
Eligibility Criteria
Baseline Phase
✅ Inclusion Criteria
- Ages 21-65 years
- Smoke ≥5 cigarettes daily for past 6 months (urine cotinine concentration greater than 200 ng/mL)
- Own iPhone or Android smartphone for daily use
- Residents of Pennsylvania, New Jersey, or Delaware
- Able to read and speak English fluently
- Fully vaccinated against COVID-19
❌ Exclusion Criteria
- Concurrent enrollment in smoking cessation programs
- Plans to use nicotine substitutes within 3 months
- Urine cotinine concentration below 200 ng/mL
- Pregnancy
- Refusal to install required mobile applications
- Phone functionality inadequate for study tasks
- Inability to upload Google Timeline data
- Planned extended trips outside PA, NJ, DE during study
Intervention Phase
📊 Participation Requirements
Participants needed to respond to ≥75% of EMA prompts during the baseline period to continue to the intervention phase, and be willing to complete store visits.
Study Protocol Evolution: From study start until October 13, 2022, participants were excluded during the intervention period for non-compliance. On October 13, 2022, the study team moved to an intent-to-treat approach and did not exclude participants due to non-compliance once they began the intervention period.
fMRI Component
🧠 Neuroimaging Session
Participants who completed the baseline and intervention study components were eligible for the optional fMRI component, subject to additional screening criteria:
Medical & Substance Use Exclusions
- Urine cotinine concentration below 200 ng/mL at scanning session
- Current or recent (within 5 years) medical treatment for substance use disorder
- Use of specified drugs within 2 weeks or plans to use within 6 weeks: Benzodiazepines, Amphetamines, Methamphetamines, Cocaine, MDMA, Methadone, Barbiturates, PCP, Heroin, Oxycodone, Opiates, Buprenorphine
- Positive drug test at scan appointment
Neurological & Psychiatric Exclusions
- Schizophrenia or psychosis (regardless of treatment status)
- History of stroke or neurological disorder affecting cognition
- Psychiatric hospitalization within the past year
MRI Safety & Scheduling Exclusions
- Propensity to experience claustrophobia
- Ferromagnetic metal in the body
- Metal in the body of unverifiable origin
- Non-removable piercings or dental work incompatible with fMRI
- Orthopedic implants above the neck
- Weight exceeding 350 pounds
- Any medical condition compromising participant safety
- Must be able to schedule scan within 6 months after completing the third Online Session
Recruitment
📢 Primary Recruitment Strategy
Recruitment materials for this study were primarily distributed by BuildClinical, utilizing study advertisements to engage participants on digital platforms such as Facebook, Google, and WebMD.
Recruitment Process
All recruitment materials included links (e.g., URLs, QR codes) directing potential participants to the study landing page at buildclinical.com/study/upenn-falk-smoking, which then directed them to complete the initial screening survey (Screen A) on the BuildClinical platform.

🔄 Data Flow Process
- Information collected via BuildClinical
- Transferred securely to REDCap
- Eligible participants automatically identified
- Participants notified via text and email
- Invited to schedule phone screening
👥 Referral Process
- Some participants requested referral links
- Direct link provided: redcap.link/georemote
- Participants asked not to discuss study details
- Privacy maintained until both completed participation
fMRI Recruitment: Enrolled or completed participants who were potentially eligible for the optional fMRI session were contacted to screen them for interest in and eligibility for the scan session.
Consent
📋 Multi-Stage Consent Process
Informed consent was collected at multiple stages throughout the study to ensure participants were fully informed at each phase of their involvement.
Consent Timeline
Screening Consent
Electronic consent form for screening collected during 'Screen B'
Full Study Consent
Electronic consent for full study participation collected during Online Session 1
fMRI Consent
Electronic consent for fMRI component collected at beginning of fMRI screening survey and scan session
Consent Details
Screening Phase: Participants eligible following Screen B were directed to Online Session 1, where they provided electronic consent to participate in the full study.
fMRI Component: Participants who completed the baseline and intervention components and who were interested in the fMRI component provided electronic consent at the beginning of the fMRI screening survey. Eligible participants who completed the fMRI scan provided additional consent at the beginning of the session.
Screening and Enrollment Process
📝 Screen A
Those who expressed interest in the study were directed toward an online screening survey (Screen A), administered via BuildClinical or REDCap.
Screen A Components:
- Questions relevant to study eligibility
- Contact information collection
- Interest in future study opportunities
- Duration: Less than 5 minutes
Follow-up Process:
- Ineligible individuals were informed via email
- Potentially eligible participants received email and text with Calendly link to schedule initial screening phone call
Initial Screening Call
📞 Call Preparation
- Participants emailed copy of consent form
- Study timeline provided for review
- Scheduled via Calendly appointment system
🗣️ Call Components
- Detailed study information provided
- Risks and benefits discussion
- Q&A opportunity
- Eligibility criteria confirmation
Next Steps: Eligible and interested participants were invited via email to complete Screen B within 1 month of the initial call.
🔐 Screen B
Screen B began with an electronic consent form for the online screening survey and included several technical and verification components.
Required Components
Documentation:
- Shipping address provision
- COVID-19 vaccination card photo submission
Technical Setup:
- Google Maps download and setup with correct location tracking settings
- Google Timeline (location history) data export and upload
- Detailed instructions provided for both iPhone and Android systems
- Optional technical support via phone call with experimenter
COVID-19 Precautions: Vaccination was required because the pandemic was classified as a federal public health emergency when the study began, with extra precautions implemented to reduce risks during retail store visits.
Verification Process
Location Verification:
- Confirmed participants lived in study area (PA, NJ, or DE) using uploaded timeline data and shipping address
Vaccination Verification:
- Confirmed full COVID-19 vaccination status using uploaded vaccination card
Exclusion Criteria at Screen B:
- Unable or unwilling to complete any Screen B component
- Lived outside specified study area (PA, NJ, DE)
- Not fully vaccinated against COVID-19
Data Protection: If individuals were found ineligible, the team informed them of ineligibility, deleted their geolocation data, and provided instructions for turning off location tracking and uninstalling Google Maps if desired.
📦 Mailing Materials
Eligible individuals were mailed a comprehensive box containing all necessary study materials.
Mailed Materials Included:
- Urine cotinine test
- Greenphire Clincard for study payments
- KN95/KF94 or N95 masks (for COVID-19 protection during retail store visits)
Upon Receipt: Participants received instructions to enroll in the study and begin participation by completing Online Session 1.
🖥️ Online Session 1
Comprehensive session administered over REDCap and Qualtrics, expected to take 30-60 minutes.
Session Components
Consent and Payment:
- Electronic consent via REDCap for full study participation
- Study payment information collection
Assessments:
- Self-report measures completion
- Physiological measurements (urine cotinine) to confirm smoking status
EMA Training:
- Instructions for Baseline period EMA task
- Comprehension assessment questions
- RealLife Exp app installation, setup, and practice
Cotinine Verification
Final Eligibility Check: Participants uploaded a photograph of their urine cotinine test result. If results were not above the required threshold, participants were excluded from continuing in the study.
Initial Interest
Online screening survey (<5 min)
Detailed Screening
Phone consultation with researcher
Technical Setup
Location tracking, vaccination verification
Study Kit
Cotinine test, payment card, masks
Full Enrollment
Consent, training, final verification
Baseline Period
📊 Baseline Data Collection
The baseline period began 2 days after completion of Online Session 1, and lasted for 14 days. During this period, participants completed EMA and location tracking to establish baseline smoking patterns.
Baseline Requirements
Duration: 14 days starting 2 days after Online Session 1
Data Collection:
- EMA surveys
- Continuous geolocation tracking for tobacco retail exposure assessment
Quality Assurance
📞 Participant Support
- Participants contacted if no EMA surveys completed in first few days
- Regular updates of % of surveys completed
- Technical support as needed
🎯 Compliance Requirements
- ≥75% EMA response rate required
- Geolocation tracking must be active (verified through location history files)
Flexibility: Participants were able to pause their participation as needed (for example, due to significant illness or disruptive life events) at any point during the study.
Online Session 2
🖥️ Pre-Intervention Session
Participants completed Online Session 2 after the baseline period, with encouragement to complete within 1 week of invitation.
Session Components
Duration: 30-60 minutes via Qualtrics
Tasks Completed:
- Self-report measures
- Image rating task
- Geolocation data export and upload
- EMA setup and practice
- Intervention task instructions and practice
Image Rating Task
🖼️ Smoking Cue Reactivity Assessment
Participants viewed photographs and rated their craving response to assess reactivity to different types of smoking and retail cues.
Task Structure:
- 35 image sets (2 images per set)
- 3-second exposure per image
- Craving rating 0-100 scale after each set
- Technical issue reporting via checkbox option
7 Categories each repeated 5 times
Categories 1-2: Standard Cues
1. Smoking cues: Cigarettes in a pack, in an ashtray, or alongside a lighter
2. Non-smoking cues: Pencils in a pack, with sharpener, or sitting on a table
Categories 3-4: Retail Environments
3. Non-tobacco retailers: Cash register areas at non-tobacco retailer stores
4. Tobacco retailers: Cash register and power wall area at tobacco retailer stores
Categories 5-7: Product Images
5. Cigarette packs: Close-up photographs of single brand cigarette packs
6. Promoted cigarettes: Single brand cigarette packs with price promotion features
7. Control products: Close-up photographs of gum packs with price promotion features
Personalization Features:
- Filter customization: Brown or white filters matched to participant preferences
- Brand customization: L&M, Pall Mall, Camel, Newport, or Marlboro based on preference
- Randomized presentation: Category order randomized, image pairs within categories
Pre-Task Assessment:
- Time since last cigarette
- Current craving level
- Instruction to refrain from smoking during task
Additional Eligibility Screening
Store Visit Capability: Starting 10/06/2023, participants were asked if they could complete store visits (before knowing their assignment). Those unable to complete visits were excluded and compensated for completed tasks.
Random Condition Assignment
🎲 Randomization Procedure
Participants who completed baseline requirements were randomized using a blocked design to ensure balanced group assignment.
Randomization Strategy
Blocking Variables:
- Gender: Male, Female, Other
- Smoking level: High (≥20 cigarettes/day), Low (<20 cigarettes/day)
Implementation Timeline:
- Pre-12/01/2022: Fully random assignment (66 participants)
- Post-12/01/2022: Blocked randomization (216 participants)
Study Conditions
Tobacco Retailer
Enter 7-11 stores that sell tobacco within study area (PA, DE, NJ)
Non-Tobacco Retailer
Enter CVS stores that do not sell tobacco within study area
Control
No store entry required, equal compensation, same other tasks
Intervention Period
🏪 4-Week Intervention Phase
The intervention period began after Online Session 2 completion, involving 4 weeks of condition-specific store visits alongside continued EMA and location tracking.
Universal Tasks (All Participants)
Duration: 4 weeks
Required Activities:
- EMA surveys
- Continuous location tracking
- Weekly surveys
Store Visit Conditions
🏪 Store Visit Requirements
- Frequency: 5 times per week for 4 weeks
- Purchase requirement: Small purchase (excluding tobacco)
- Budget: $3 per visit via Greenphire Clincard
- Documentation: Receipt photo via RealLife Exp app
🎯 Store Assignments
- Tobacco Retailer: Convenience stores selling tobacco
- Non-Tobacco Retailer: Pharmacy stores not selling tobacco
- Control: No store visits required
- Compensation: Equal across all conditions
Online Session 3
📋 Post-Intervention Assessment
Following the Intervention Period, participants completed Online Session 3, encouraged to complete within 1 week of invitation. Two participants completed after longer delays.
Session Components
Tasks Completed:
- Self-report surveys
- Image rating task (repeat administration)
- Geolocation data export and upload
Study Completion Procedures
📱 App Removal Instructions
- Smartphone application uninstallation guidance
- Location tracking deactivation instructions
- Participant choice for data retention
📧 Non-Responder Protocol
- Final payment sent after several contact attempts
- Smoking cessation information provided
- Study debriefing materials sent
Debriefing and Resources
Study Completion: Participants not interested in or eligible for fMRI received debriefing forms detailing study procedures and research goals, plus smoking cessation assistance information.
fMRI-Eligible Participants: Debriefing and cessation resources provided after scan completion or upon determination of scan ineligibility.
fMRI Session
🧠 Optional Neuroimaging Component
Participants who completed Online Session 3 and met eligibility criteria were invited for an optional 2-hour in-person fMRI session at the University of Pennsylvania.
Pre-Scan Procedures
Eligibility Confirmation:
- fMRI screening survey completion
- Criteria verification (as described in Eligibility section)
- Session scheduling at University of Pennsylvania
Day of Scan Protocol:
COVID-19 & Safety
University/hospital screening requirements, consent review
Final Verification
Urine sample, forms completion, surveys
fMRI Session
Instructions, training, 1-hour scan protocol
fMRI Image Rating Task
🖼️ Neuroimaging Task Parameters
Enhanced version of the online image rating task, optimized for fMRI data collection.
Task Structure:
- 70 blocks of 4 images each
- 4-second exposure per image
- 3-second response window for craving rating
- Rating scale: 1 (not at all) to 5 (very much)
Image Categories (Same 7 as Online Task):
Repetition Structure
10 repetitions per category (vs. 5 in online task)
Each image repeated twice (vs. unique images online)
Categories 1-4
1. Smoking cues
2. Non-smoking cues
3. Non-tobacco retailers
4. Tobacco retailers
Categories 5-7
5. Cigarette packs
6. Promoted cigarettes
7. Control products (gum)
Additional Scanning Components
Anatomical Scans:
- T1-weighted anatomical scan
- T2-weighted anatomical scan
- Fieldmap scan for distortion correction
Technical Details: Complete scanning parameters can be found in Appendix: fMRI Scanning Parameters.
Session Summary
Session Breakdown:
- Pre-scan procedures: COVID screening, consent, eligibility verification
- Task preparation: Safety instructions, task training
- Scanning time: ~1 hour (task + anatomical scans)
- Post-scan: Debriefing and resource provision